As the world strives to put the pandemic behind us, a burden of uncertainty continues to weigh on the minds of patients, physicians, and scientists. How durable is the immunity produced by vaccines or infection with SARS-CoV-2? To what degree does immunity extend to new variants?
The answers to these questions and more lie in a deeper understanding of the T-cell response to the virus.
T cells provide both the brain and the brawn that make our adaptive immune system so powerful. They are the first responders of the adaptive immune system and activate antibody responses. But they are underappreciated because they are more difficult to measure than antibodies.
What can T cells tell us about vaccine efficacy in the presence of variants?
Many of the variants make it easy for the virus to evade antibodies induced by the vaccines currently on the market1. In theory this is dangerous, but across the board, all of the vaccines are providing strong efficacy even in the presence of variants—even, when the level of neutralizing antibodies is much lower. Why? The leading hypothesis is that T-cell response is not significantly affected by the variants.
SARS-CoV-2 variants escape neutralization by vaccine-induced humoral immunity
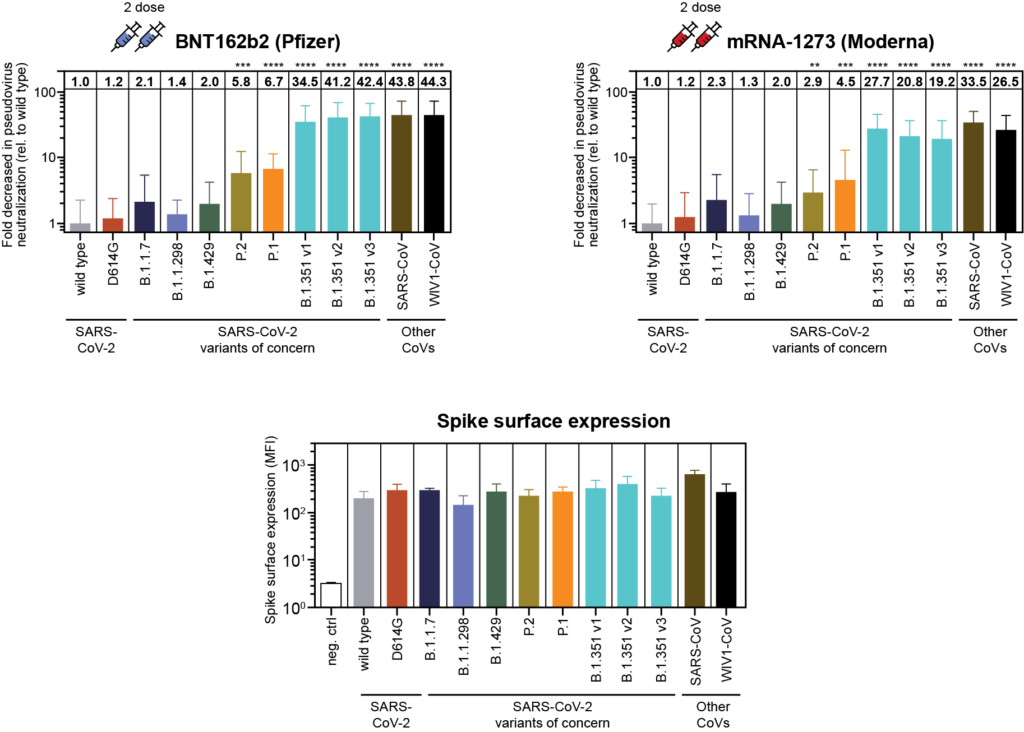
Antibodies work by binding to the virus itself and preventing it from entering cells. If the part of the virus to which the antibody binds is altered, its ability to “hang on” and thus prevent it from entering a cell is diminished. One big advantage of T cells is that they are highly specific and react to a much broader portion of the virus than antibodies, including areas outside of the spike protein. This is why T cells might provide protection against the variants, even when neutralizing antibody levels are low.
It turns out that to be protected against the COVID-19 virus, you don’t necessarily need a whopping neutralizing antibody response, you need a whopping adaptive immune response – and that includes both antibodies and T cells.
How do you measure the T-cell response?
T-cell research offers additional rich insights into immunity, creating a clearer picture of the total immune response. However, there are technical hurdles in measuring T cells accurately, and until recently, it wasn’t possible to to measure T cells at a population level. This is exactly the problem we at Adaptive Biotechnologies set out to solve. Thanks to advanced sequencing, machine learning and cloud computing, we are now able to do this research with precision and scale never before possible.
Adaptive’s proprietary research and data analysis service, immunoSEQ® T-MAP COVID, combines the sequencing and mapping capabilities of our immune medicine platform to map T-cell receptors (TCRs) to disease-specific antigens. Over the course of the last year, we have acquired more than 6,000 patient samples to identify TCRs to all parts of the virus. Using this tool, we have been able to show how T cells respond to different parts of the virus, including the different parts of the spike protein. And, we have even been able to show exactly how the variants impact these parts of the virus, which can help us understand whether the immune response is likely to be affected. From the research done to date, we’ve proven that the most common parts to which T cells respond are not altered by mutations from the two most common viral strains, B.1.1.7 (UK) and B.1.351 (SA) (see chart below).
Vaccine-induced CD8+ T-cell Responses to Spike Protein Antigens
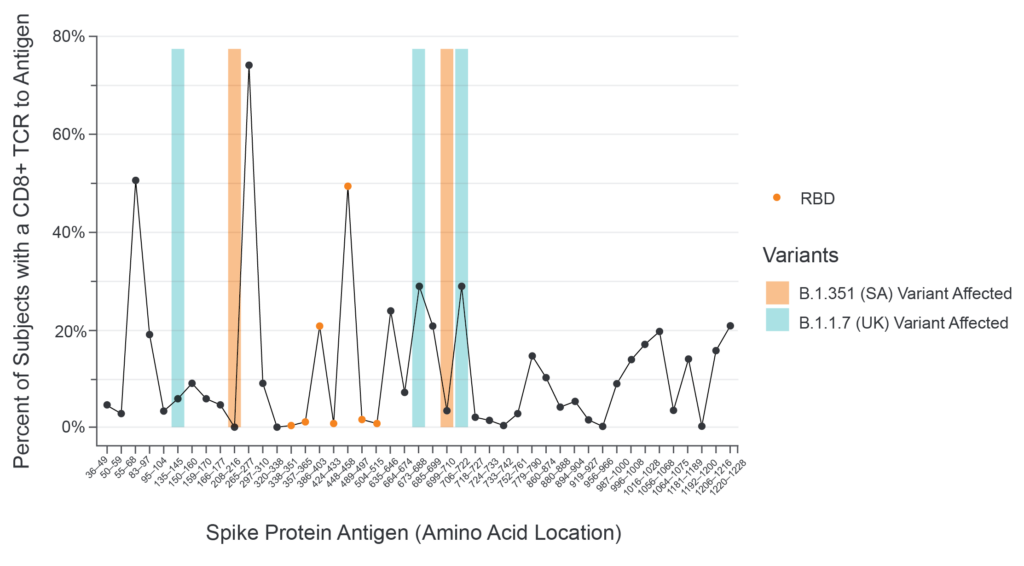
What makes this relevant in the real world? In peer-reviewed, published studies, it has been demonstrated that both the Astra Zeneca2 and Johnson and Johnson3 vaccines generate a broader cellular immune response against regions of the spike protein not impacted in any of the emerging variants, and are therefore very likely to contribute to protection from COVID-19.
What does this mean for determining immunity against the virus?
The development of vaccines against SARS-CoV-2 will go down as one of the greatest scientific and medical achievements in history. Tremendous progress has been made, and to sustain our success and re-entry into normal life, it is critical not to let our guard down.
More research is needed to clearly define and quantify T-cell-driven immunity so that we can know who is well-protected and who isn’t—at the individual and population level. As this virus becomes endemic in the population, as previous pandemic-causing pathogens have done, industry has already made a head start on next-generation vaccines. We need a reliable, sustainable, and quantitative way to measure the longevity of efficacy of the vaccines, and as we are seeing, the answers lie within the T cells.
To win the fight against COVID-19, it’s essential to understand the correlation between different markers of immunity and health outcomes. We are learning T cells can tell us more about immunity than antibodies, and immunoSEQ® T-MAP™ COVID is one of the most powerful tools available for probing the T-cell response.
1 Garcia-Beltran, et al. Multiple SARS-CoV-2 variants escape neutralization by vaccine-induced humoral immunity. Cell 2021.
2 Madhi et al. Efficacy of the ChAdOx1 nCoV-19 Covid-19 Vaccine against the B.1.351 Variant. NEJM 2021.
3 Barouch et al. Immunogenicity of Ad26.COV2.S Against SARS-CoV-2 Variants in Humans. Nature 2021.
